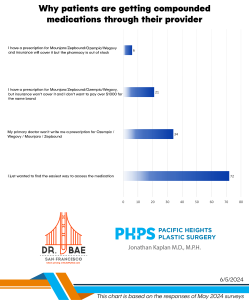
We’ve received questions about how we’re getting semaglutide, the active ingredient in Ozempic and Wegovy, since it’s currently so backordered.
We’re ordering our semaglutide from a compounding pharmacy. Never heard of a compounding pharmacy? That’s ok, chances are you’ve used medication from a compounding pharmacy before.
Examples of what a compounding pharmacy offers:
- Allergic to a dye or certain ingredient in a medication? A compounding pharmacy would recreate that medication without the dye or allergen
- Have a child, or elderly parent that can’t swallow the pill form of a medication? A compounding pharmacy can recreate the medication in liquid form
- Ever received botox or filler at a doctor’s office? The topical numbing cream placed on the skin prior to treatment was made at a compounding pharmacy
- On bioidentical hormone replacement therapy (BHRT)? From a compounding pharmacy.
- Medications that are in short supply, including medication for COVID, can be produced by compounding pharmacies too
This last example is why we’re able to get semaglutide. It’s on the FDA’s shortage list, and as such, the FDA allows a compounding pharmacy to duplicate a commercially sold drug that’s currently unavailable or in short supply.
So what if semaglutide is removed from the shortage list? There are two schools of thought on this point.
If brand name medications are taken off the FDA shortage list, it’s possible compounding pharmacies will no longer be allowed to manufacture semaglutide. The other school of thought is that because most compounding pharmacies are combining B-12 or L-carnitine with their sterile compounded semaglutide, they are making a materially different compound than the patented version of semaglutide manufactured by NovoNordisk. And as such, compounding pharmacies can continue producing a novel version of semaglutide with this additive.
Which school of thought will win out? The FDA has not weighed in on this question yet.
Generic vs “Generic”
Also, we may use the term generic when referring to our compounding pharmacy version of semaglutide. We are aware that generic within the context of pharmaceuticals can mean a drug that is being manufactured after a previous name-brand drug has lost its patent protection. That’s certainly not the case here because Ozempic and Wegovy have not lost their patent protection.
But we will sometimes reflexively refer to our semaglutide as generic because it is 1) an alternative way of saying the medication is not a name brand, which is certainly generic in some contexts and 2) less confusing for most patients. Regardless of whether we use the term generic or replicated or analogue, it is the same active ingredient whether it was created at a compounding pharmacy or at a large pharmaceutical company that makes “generic” drugs.
Quality standards of compounding pharmacies
In regards to the quality of medications produced by a compounding pharmacy, the facility we get semaglutide from is considered an “outsourcing facility,” as defined in The Drug Quality and Security Act, of 2013. That law created a new section, 503B, in the Federal Food, Drug, and Cosmetic Act. As such, an outsourcing facility is overseen and inspected by the FDA, along with state regulators.
Under the law, outsourcing compounding pharmacies must comply with section 503B and current good manufacturing practice (CGMP) requirements. In other words, they have very high standards to keep.
And we’ve had a great experience with this compounding pharmacy providing our semaglutide. It’s a relationship that predates this particular drug. So for now, we have a robust supply of semaglutide and will maintain at least a 6 month availability while supplies last.
If you’re not in our program, read more about semaglutide here and prioritize scheduling your consultation by paying online here.